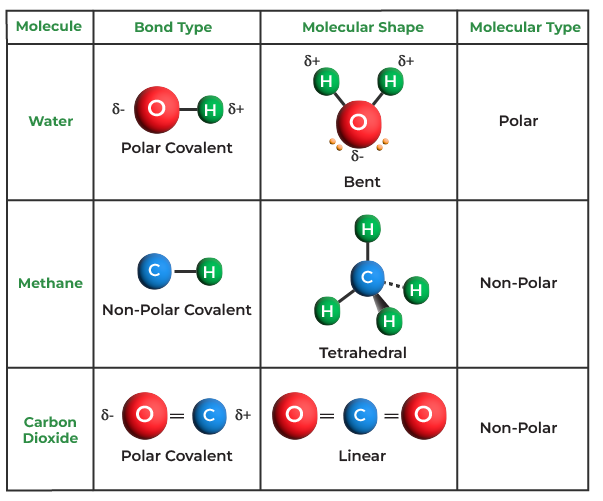
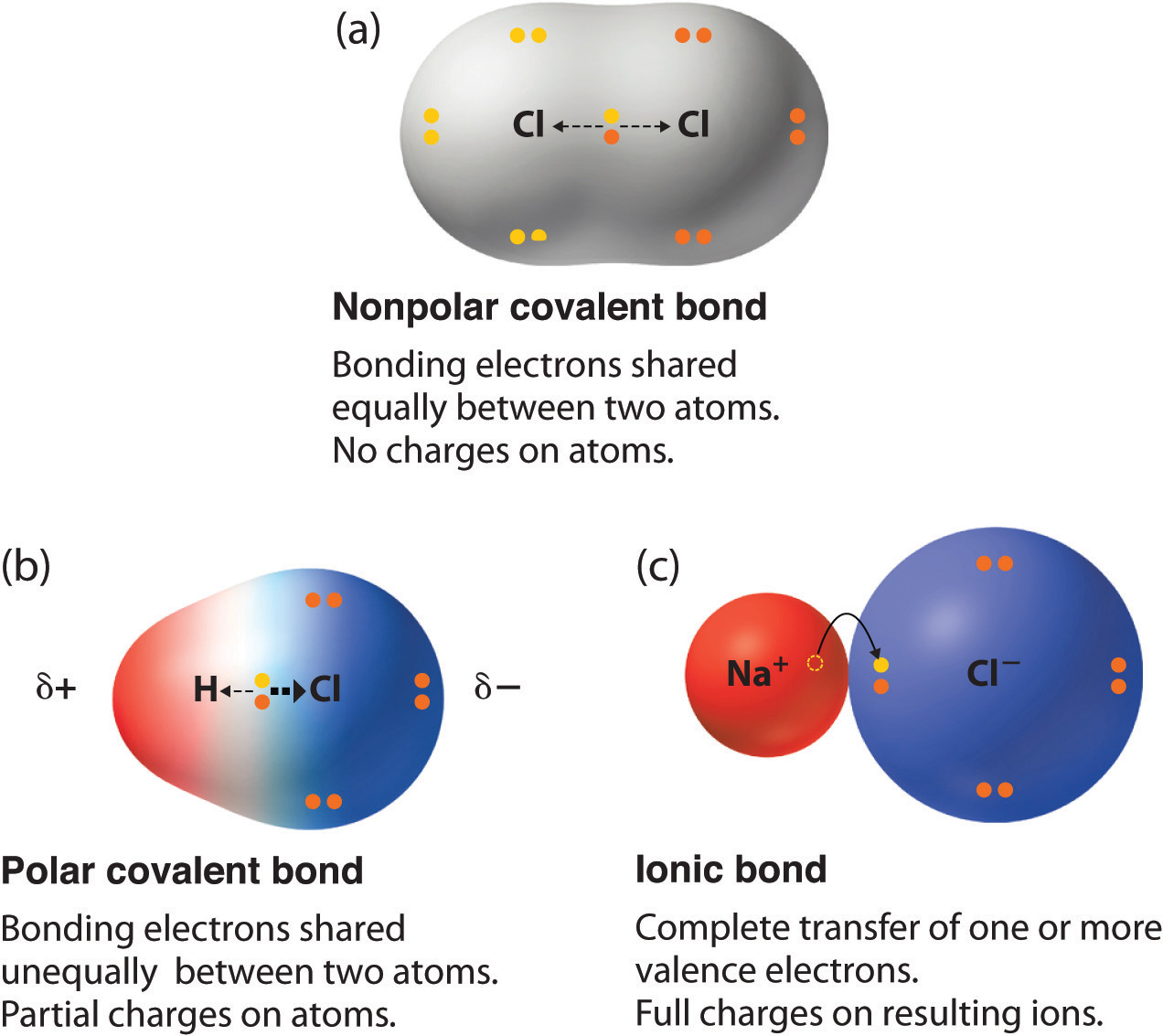
Which Combination Of Atoms Can Form A Polar Covalent Bond - This occurs when one atom has a higher. एक कार्ब अणू __________ सहसंयुज बंध तयार करू शकतो. Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule are polar. Only h and br form polar covalent bond. A polar covalent bond is created when the shared electrons between atoms are not equally shared. As demonstrated below, bond polarity is a useful concept for describing the sharing of electrons between atoms, within a. Covalent bonding occurs when pairs of electrons are shared by atoms.
एक कार्ब अणू __________ सहसंयुज बंध तयार करू शकतो. As demonstrated below, bond polarity is a useful concept for describing the sharing of electrons between atoms, within a. This occurs when one atom has a higher. A polar covalent bond is created when the shared electrons between atoms are not equally shared. Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule are polar. Covalent bonding occurs when pairs of electrons are shared by atoms. Only h and br form polar covalent bond.
Only h and br form polar covalent bond. Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule are polar. Covalent bonding occurs when pairs of electrons are shared by atoms. A polar covalent bond is created when the shared electrons between atoms are not equally shared. This occurs when one atom has a higher. एक कार्ब अणू __________ सहसंयुज बंध तयार करू शकतो. As demonstrated below, bond polarity is a useful concept for describing the sharing of electrons between atoms, within a.
Polar Covalent Bonds Electronegativity
A polar covalent bond is created when the shared electrons between atoms are not equally shared. As demonstrated below, bond polarity is a useful concept for describing the sharing of electrons between atoms, within a. Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule are polar. This occurs when one atom has a.
Reading Covalent Bonds Biology I
Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule are polar. Covalent bonding occurs when pairs of electrons are shared by atoms. A polar covalent bond is created when the shared electrons between atoms are not equally shared. Only h and br form polar covalent bond. एक कार्ब अणू __________ सहसंयुज बंध तयार.
Definition and Examples of a Polar Bond
As demonstrated below, bond polarity is a useful concept for describing the sharing of electrons between atoms, within a. Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule are polar. This occurs when one atom has a higher. एक कार्ब अणू __________ सहसंयुज बंध तयार करू शकतो. A polar covalent bond is created.
II. Kinds of Chemical Bonds Ionic Bond Covalent Bond Comparison Chart
This occurs when one atom has a higher. Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule are polar. A polar covalent bond is created when the shared electrons between atoms are not equally shared. Covalent bonding occurs when pairs of electrons are shared by atoms. Only h and br form polar covalent.
Polar Covalent Bond Definition And Examples, 49 OFF
A polar covalent bond is created when the shared electrons between atoms are not equally shared. As demonstrated below, bond polarity is a useful concept for describing the sharing of electrons between atoms, within a. एक कार्ब अणू __________ सहसंयुज बंध तयार करू शकतो. Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule.
Covalent Bond Definition and Examples
A polar covalent bond is created when the shared electrons between atoms are not equally shared. Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule are polar. As demonstrated below, bond polarity is a useful concept for describing the sharing of electrons between atoms, within a. Covalent bonding occurs when pairs of electrons.
Polar Covalent Bonds Clearly Explained for Easy Learning
Covalent bonding occurs when pairs of electrons are shared by atoms. As demonstrated below, bond polarity is a useful concept for describing the sharing of electrons between atoms, within a. Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule are polar. Only h and br form polar covalent bond. एक कार्ब अणू __________.
SOLVED In a polar covalent bond, A) both atoms in the bond have the
As demonstrated below, bond polarity is a useful concept for describing the sharing of electrons between atoms, within a. Covalent bonding occurs when pairs of electrons are shared by atoms. एक कार्ब अणू __________ सहसंयुज बंध तयार करू शकतो. This occurs when one atom has a higher. Only h and br form polar covalent bond.
Chapter 5.6 Properties of Polar Covalent Bonds Chemistry LibreTexts
Only h and br form polar covalent bond. Covalent bonding occurs when pairs of electrons are shared by atoms. As demonstrated below, bond polarity is a useful concept for describing the sharing of electrons between atoms, within a. एक कार्ब अणू __________ सहसंयुज बंध तयार करू शकतो. This occurs when one atom has a higher.
Solved Question 1 (1 point)A polar covalent bond would form
Only h and br form polar covalent bond. A polar covalent bond is created when the shared electrons between atoms are not equally shared. एक कार्ब अणू __________ सहसंयुज बंध तयार करू शकतो. This occurs when one atom has a higher. Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule are polar.
This Occurs When One Atom Has A Higher.
A polar covalent bond is created when the shared electrons between atoms are not equally shared. As demonstrated below, bond polarity is a useful concept for describing the sharing of electrons between atoms, within a. Covalent bonding occurs when pairs of electrons are shared by atoms. Explain why the covalent bonds between , oxygen and hydrogen atoms in h 2 o molecule are polar.
एक कार्ब अणू __________ सहसंयुज बंध तयार करू शकतो.
Only h and br form polar covalent bond.


/PolarConvalentBond-58a715be3df78c345b77b57d.jpg)