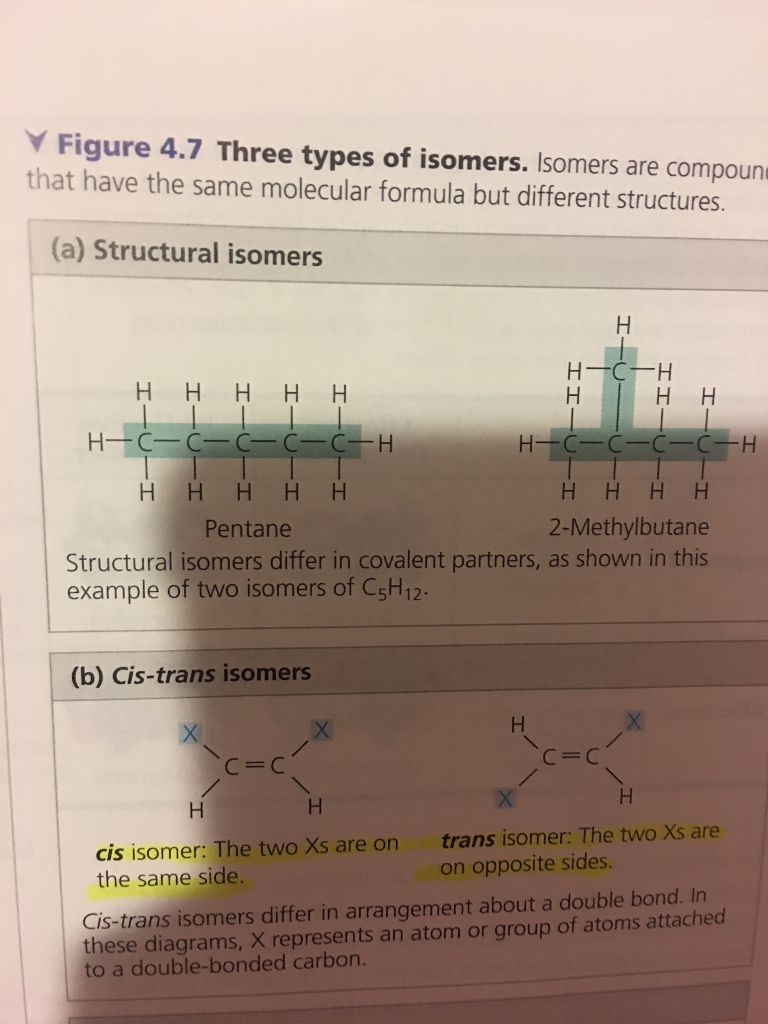
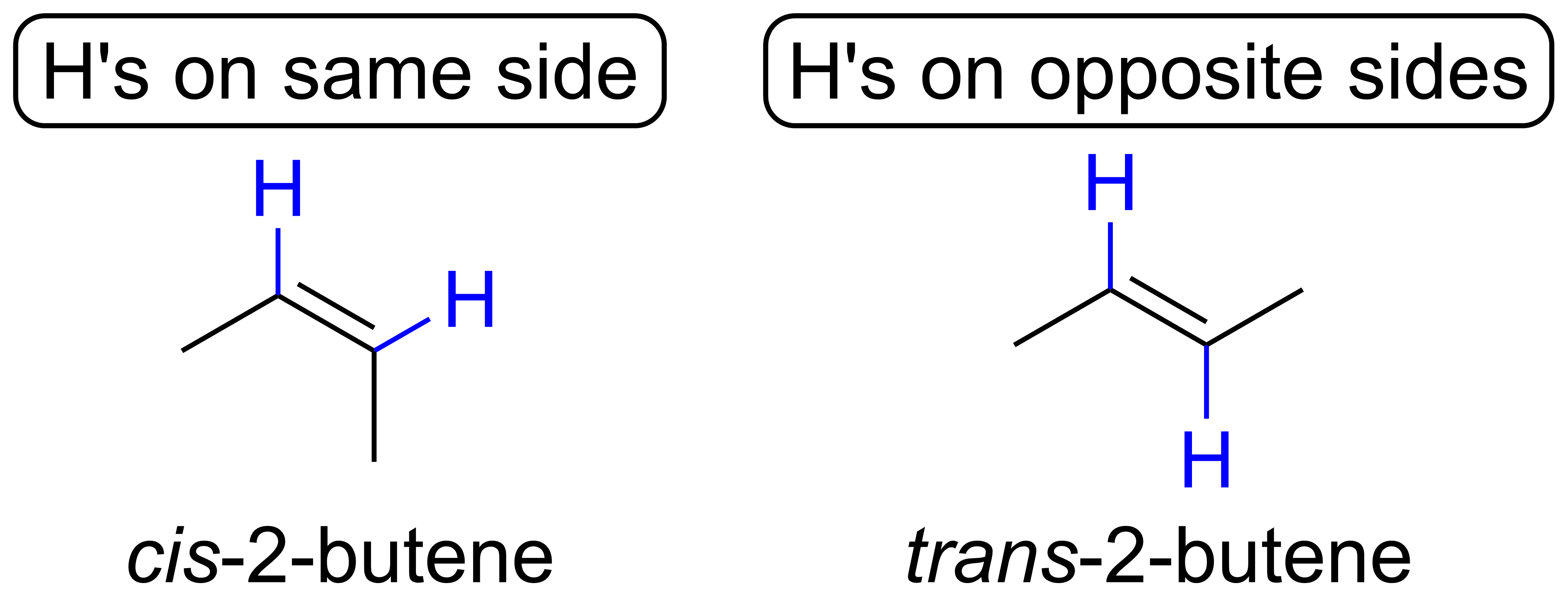
Can Propane Form Isomers - Find out why propane cannot have any isomers due to its. Learn the definition and examples of structural isomerism in organic chemistry. As there is no double bond in. For isomers to exist, there should be a minimum of four carbons for branching to take place. Propane cannot form structural isomers because the three carbons atoms can attach only in a straight line. Propane (c3h8) does not have any structural isomers. Structural isomers are compounds with the same molecular formula but different. However iff there is hetero atom or any atom. In alkanes, isomerism arises when a particular compound.
Structural isomers are compounds with the same molecular formula but different. Propane cannot form structural isomers because the three carbons atoms can attach only in a straight line. Find out why propane cannot have any isomers due to its. As there is no double bond in. For isomers to exist, there should be a minimum of four carbons for branching to take place. Propane (c3h8) does not have any structural isomers. In alkanes, isomerism arises when a particular compound. However iff there is hetero atom or any atom. Learn the definition and examples of structural isomerism in organic chemistry.
For isomers to exist, there should be a minimum of four carbons for branching to take place. As there is no double bond in. In alkanes, isomerism arises when a particular compound. Propane cannot form structural isomers because the three carbons atoms can attach only in a straight line. However iff there is hetero atom or any atom. Propane (c3h8) does not have any structural isomers. Find out why propane cannot have any isomers due to its. Structural isomers are compounds with the same molecular formula but different. Learn the definition and examples of structural isomerism in organic chemistry.
Una Breve Guida per Tipi di Isomeria in Chimica Organica Interesse
For isomers to exist, there should be a minimum of four carbons for branching to take place. Propane cannot form structural isomers because the three carbons atoms can attach only in a straight line. Find out why propane cannot have any isomers due to its. However iff there is hetero atom or any atom. Structural isomers are compounds with the.
Chapter 4 Carbon and its Compounds NCERT Solutions for Class 10
Structural isomers are compounds with the same molecular formula but different. For isomers to exist, there should be a minimum of four carbons for branching to take place. Propane (c3h8) does not have any structural isomers. However iff there is hetero atom or any atom. As there is no double bond in.
How many structural isomers can you draw pentane?
Propane (c3h8) does not have any structural isomers. As there is no double bond in. In alkanes, isomerism arises when a particular compound. Learn the definition and examples of structural isomerism in organic chemistry. Structural isomers are compounds with the same molecular formula but different.
CHEM Alkanes and Alkenes chemistry organic chemistr...
In alkanes, isomerism arises when a particular compound. Learn the definition and examples of structural isomerism in organic chemistry. However iff there is hetero atom or any atom. Propane cannot form structural isomers because the three carbons atoms can attach only in a straight line. Structural isomers are compounds with the same molecular formula but different.
SOLVED What are isomers? Write the isomers of butane and give their
For isomers to exist, there should be a minimum of four carbons for branching to take place. Structural isomers are compounds with the same molecular formula but different. Propane cannot form structural isomers because the three carbons atoms can attach only in a straight line. Propane (c3h8) does not have any structural isomers. Learn the definition and examples of structural.
SOLVED 1) Draw structural formula for C2H4. 2) How are gasoline and
Propane (c3h8) does not have any structural isomers. Find out why propane cannot have any isomers due to its. However iff there is hetero atom or any atom. Learn the definition and examples of structural isomerism in organic chemistry. Structural isomers are compounds with the same molecular formula but different.
Solved See figures 4.5a and 4.7. Can propane (C3H8) form
Propane cannot form structural isomers because the three carbons atoms can attach only in a straight line. Learn the definition and examples of structural isomerism in organic chemistry. Propane (c3h8) does not have any structural isomers. Find out why propane cannot have any isomers due to its. In alkanes, isomerism arises when a particular compound.
Isomers vs Conformers CHEM123 chirp
In alkanes, isomerism arises when a particular compound. Propane cannot form structural isomers because the three carbons atoms can attach only in a straight line. For isomers to exist, there should be a minimum of four carbons for branching to take place. Find out why propane cannot have any isomers due to its. As there is no double bond in.
Why are there no isomers of propane?
However iff there is hetero atom or any atom. Propane cannot form structural isomers because the three carbons atoms can attach only in a straight line. As there is no double bond in. Structural isomers are compounds with the same molecular formula but different. Find out why propane cannot have any isomers due to its.
What Are The Isomers Of Propanol? How Are They Determined?
Structural isomers are compounds with the same molecular formula but different. For isomers to exist, there should be a minimum of four carbons for branching to take place. Propane (c3h8) does not have any structural isomers. Propane cannot form structural isomers because the three carbons atoms can attach only in a straight line. As there is no double bond in.
Propane (C3H8) Does Not Have Any Structural Isomers.
As there is no double bond in. Propane cannot form structural isomers because the three carbons atoms can attach only in a straight line. Learn the definition and examples of structural isomerism in organic chemistry. Find out why propane cannot have any isomers due to its.
For Isomers To Exist, There Should Be A Minimum Of Four Carbons For Branching To Take Place.
Structural isomers are compounds with the same molecular formula but different. However iff there is hetero atom or any atom. In alkanes, isomerism arises when a particular compound.